Data integrity is important whatever line of work you’re in, but within the world of pharmaceutical manufacturing ensuring data integrity is a key component to complying with globally recognised standards.
Title 21 of the Code of Federal Regulations; Part 11, Electronic Records; Electronic Signatures (also known as 21 CFR Part 11), defines how electronic records, and associated electronic signatures on those records, can be submitted to the FDA (Food and Drug Administration). Whilst it is an American standard, it is widely followed globally, as pharmaceutical manufacturers who wish to sell into the US market will need to comply with its requirements.
Refractometer Software To Help Ensure Data Integrity
Bellingham + Stanley’s RFM900-T series of refractometers contain many software features to help ensure compliance with 21 CFR Part 11.
Subpart A of the regulation provides an overview of what the regulations apply to and provides definitions. Subpart B covers electronic records, and requirements for validation, security, authenticity of records and audit.
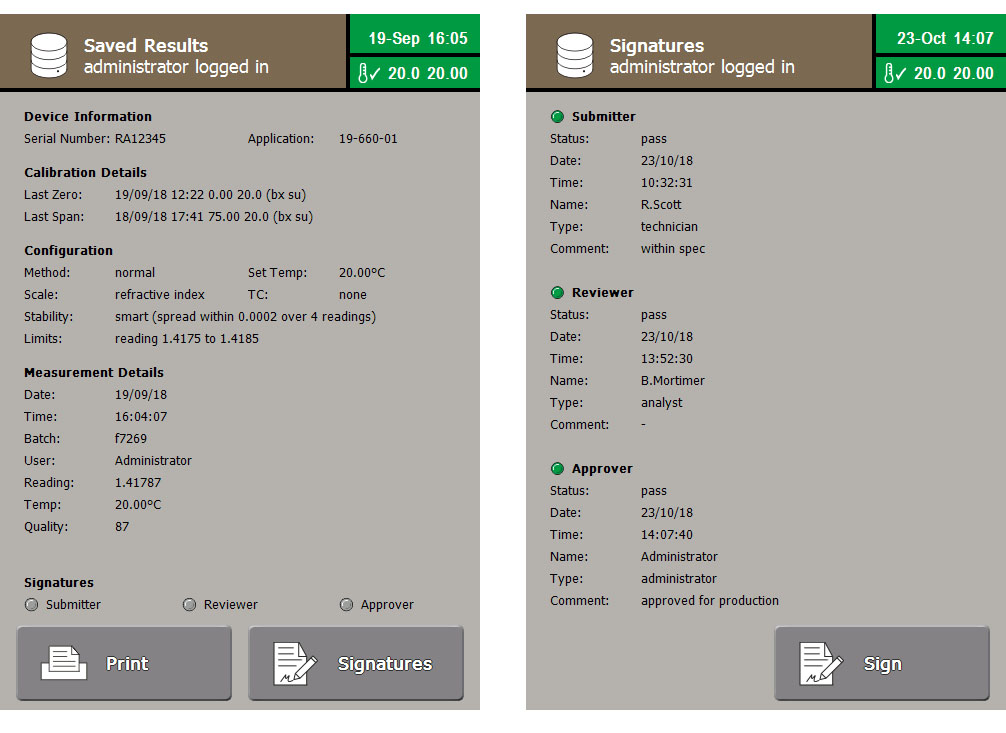 To help with this, the refractometers feature Audit trails, which cannot be switched off, capable of storing up to 10,000 entries, as well as a separate reading log for another 10,000 reading results. Before any activity is performed the user must login with their username and password, this ensures all events are recorded with the name of the person performing it as well as the time and date. Logins are kept secure by demanding minimum password lengths, customisable to local requirements, and password complexity is ensured as the system can enforce the need for combinations of upper and lower case letters, numbers and symbols.
To help with this, the refractometers feature Audit trails, which cannot be switched off, capable of storing up to 10,000 entries, as well as a separate reading log for another 10,000 reading results. Before any activity is performed the user must login with their username and password, this ensures all events are recorded with the name of the person performing it as well as the time and date. Logins are kept secure by demanding minimum password lengths, customisable to local requirements, and password complexity is ensured as the system can enforce the need for combinations of upper and lower case letters, numbers and symbols.
Security is further assured by the ability to have passwords expire after a set period, the prevention of recycling of old passwords and locking users out if they incorrectly log in too many times.
Subpart C of 21 CFR part 11 focusses on electronic signatures, their use and application and controls to manage them.
 The RFM900 series of refractometers enforces that both usernames and the signature associated with each user are unique, this ensures each signing can only be attributed to one user. When results are signed, not only is the user’s signature applied but that user’s role and the time and date of signing also recorded. Events in the system logs cannot be overwritten or altered. This ensures results are attributable, legible, contemporaneous, original and accurate (ALCOA).
The RFM900 series of refractometers enforces that both usernames and the signature associated with each user are unique, this ensures each signing can only be attributed to one user. When results are signed, not only is the user’s signature applied but that user’s role and the time and date of signing also recorded. Events in the system logs cannot be overwritten or altered. This ensures results are attributable, legible, contemporaneous, original and accurate (ALCOA).
User’s roles are fully definable, so access can be granted to any function of the instrument as applicable, whether a technician who is only given permission to perform readings, or an analyst who is granted access to the data logs to review data, whilst both are prevented from accessing system settings, such as clock adjustment or Method configuration. Their role also includes a signature level so they can sign as a submitter of a result or a supervisor can later review and approve the final results.
Easy Data Transfer From Refractometer To LIMS
Many companies will already operate a LIMS for long term storage of data. The RFM900-T series of refractometers has been designed to easily transfer data from the instrument to a LIMS.
Records on the instrument can be generated in electronic form, as well as paper based. These electronic records can be saved to the instrument’s FTP memory. By connecting the refractometer to a network via its Ethernet port the files can be easily transferred to anywhere on the network or into the LIMS. This also avoids the use of USB memory sticks which can’t be used in many companies due to their insecure nature (although a USB option is still available if wanted!).
Whenever a file is created on the instrument a checksum file is also created. This can be exported from the instrument at the same time as the main file, then using free Bellingham + Stanley software, “MD5 file verifier”, the checksum can be recalculated on the file and compared to the original thus ensuring that the file has maintained its integrity in the transfer and confirm that no one has subsequently altered it.
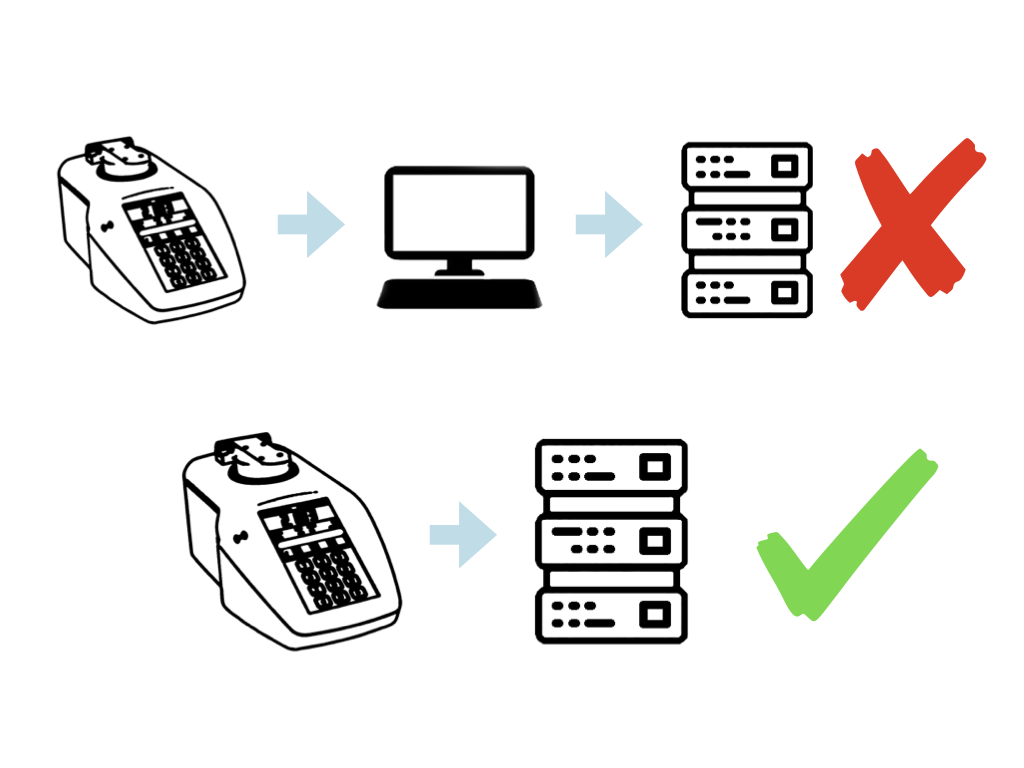 The instrument logs can be easily filtered by date range, user, signed status, Method used and export status. This way the required data can be easily found, reviewed, signed and exported to LIMS. Transfer of logs to the FTP memory can also be automated so after a period of time logs will be exported in the background without the need for an operator to be present, and with free Bellingham + Stanley software “FTP instrument sync” those files placed anywhere on a network.
The instrument logs can be easily filtered by date range, user, signed status, Method used and export status. This way the required data can be easily found, reviewed, signed and exported to LIMS. Transfer of logs to the FTP memory can also be automated so after a period of time logs will be exported in the background without the need for an operator to be present, and with free Bellingham + Stanley software “FTP instrument sync” those files placed anywhere on a network.
All file transfer is limited to users who are granted access to the rights via their user role.
Traceable Validation and Calibration From UKAS Accredited Lab
Bellingham + Stanley can also provide system validation (IQOQPQ) and traceable calibration standards from our UKAS accredited lab. With additional RFM900-T refractometer series features such as automated system backups, network user authentication and network controlled instrument time, the refractometer can provide everything needed to help operate in a 21 CFR part 11 environment.
Familiar Software Across A Range Of Products
If you don’t require the power and features of the RFM900-T Series refractometers, or are looking for the same data integrity solutions in a polarimeter then Bellingham + Stanley can help.
The software features detailed above are available on a number of Bellingham + Stanley products including the RFM300-T Series refractometers, ADP450 polarimeters, and ADP600 Series polarimeters. No matter which model you choose, the fast and easy to use software will enable you to comply to FDA regulation 21 CF part 11.
For More Information Contact Us Today
For further reading on how Bellingham + Stanley can help you comply with FDA regulations 21 CF part 11, take a look at our blog “how to comply” or head over to our channel on YouTube to watch our video on the same subject.
Ready to move to the next step? Contact us today and ask about Bellingham + Stanley polarimeters and refractometers. To read more about Bellingham + Stanley refractometers visit our Refractometer Overview page.
You can also request a customer information pack which includes instrument validation reports, software compliance reports and instrument connectivity reports but contacting our customer care team.